This post was last updated on March 28, 2026.
Increasingly in the news in North America and Europe, dengue fever is caused by a mosquito-borne virus that infects an estimated 390 million people every year. Each year it also kills many of thousands of people, leading the World Health Organization to describe it as one of the top 10 threats to global health. Yet for most people who haven’t lived or travelled to the areas it most affects, it’s an unfamiliar virus.
I am sharing resources about dengue for several reasons. First, because the virus’ footprint continues to expand as our planet’s weather patterns change and mosquito areas change with it. It is now cropping up in parts of Europe and North America, where people are less knowledgeable about the condition. Second, because as I note below its antibodies can cross-react with Covid, and Covid remains a health issue worldwide that impacts our immune systems, even if we aren’t in the early phases of a pandemic any longer.
And finally because it changed my life — and not for the better, either.

For my story of how I got dengue fever in Vietnam, please see my Legal Nomads post here.
The disease is now present in more than 130 countries per the World Health Organization, who also note that cases have surged in recent years. Their dengue dashboard allows you to track the virus worldwide. From February 2025 to February 2026, the dashboard notes a total of 5,238,017 cases and several thousand deaths.

Why the increased spread? Several factors at play, from changing distribution of the vectors themselves (primarily the Aedes aegypti and Aedes albopictus mosquitoes), as well as warming climates, record rainfall, and humidity. While the viral fever is common in parts of Asia and the Americas, global warming has affected the reach of the mosquitoes that spread it. In September 2022, for example, France sounded the alarm about an outbreak of locally-transmitted dengue.
In addition, deaths may be higher due to cross-reactivity with Covid-19 (see below), and/or the fragility of the medical systems in many countries around the world who are managing both ongoing high levels of Sars-Cov-2 infection plus underfunding or understaffing in the medical system. Perhaps people are also not realizing they have dengue, thinking they have Covid, and thus not seeking medical care when most crucial. And as countries have focused chiefly on Covid-19 the last few years, other surveillance programmes have missed funding or response support.
A study a few years ago also found that being infected with the virus causes you to produce a bacteria in the skin that makes you more attractive to mosquitoes. Not the kind of news anyone wants to read! Hopefully this team’s findings could help inform real-world public health strategies for controlling mosquito-borne flaviviruses like dengue and Zika, because by 2080, as many as 2.25 billion more people — or 60% of the global population — will be at risk.
Presently there is no cure for dengue fever, only management and prevention.
Dengue fever basics: serotypes, symptoms & immunity
There are five dengue fever serotypes – DenV 1, 2, 3, 4 and 5. The fifth serotype was only established in 2015, but manifestation of symptoms differ depending on the type you contract (source).
I was told often in Asia that recovery from one strain of dengue may offer lifelong immunity against that strain, and after two infections you may be immune to all serotypes. However, in 2023 a study from Nicaragua shed some light on this long-standing belief. The results contradicted the conventional wisdom that people can become immune to the dengue virus for the rest of their lives after repeated infections. Instead, the models from that study suggest that immunity is temporary, and wanes eventually—suggesting dengue is less like measles, and more like the flu or Covid-19.
In 2020, Singapore reported a spike in the DenV-3 type of dengue, which their National Environment Agency noted may signal an outbreak in the near future. The DenV-1 and DenV-2 serotypes were dominant in Singapore for the last three decades, so the country was on alert due to the rise of DenV-3 since population immunity is low and people are more susceptible to it.
Eradicating the mosquito-borne disease is likely an impossibility, though many scientists are racing to find solutions.
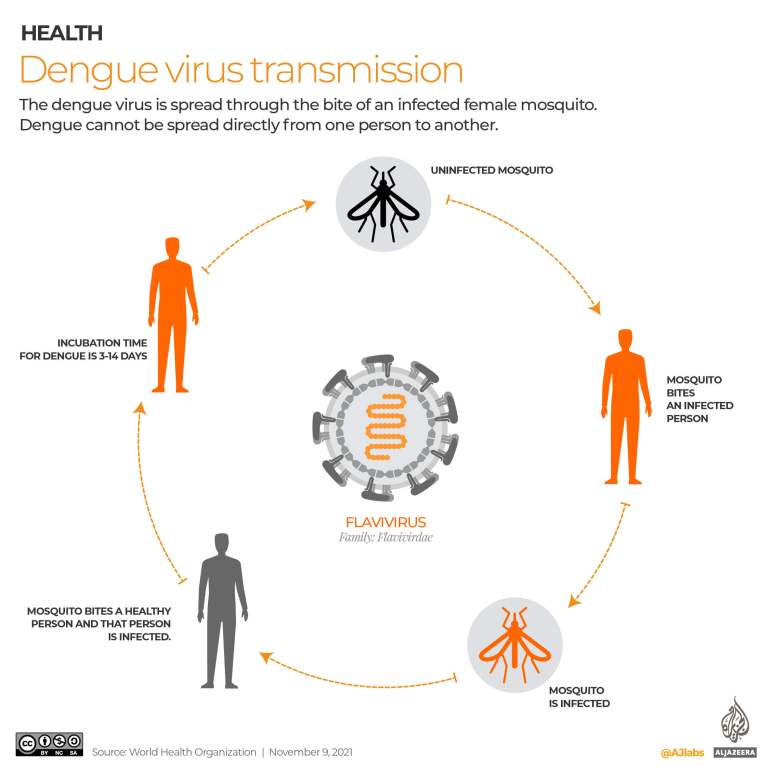
How dengue virus transmits between mosquitoes and humans. Via Al Jazeera
Dengue’s incubation period is between 4-10 days, so it’s difficult to backtrack to exactly what mosquito bit you. In my case, I have a very good idea. I was outside on a phone call and came inside so bitten up that I almost vomited. It was less than a week later that my symptoms started.
According to the CDC, the principal symptoms of dengue are high fever, feeling general malaise (like the flu), and at least two of the following:
- Severe headache
- Severe eye pain (behind eyes)
- Joint pain
- Muscle and/or bone pain
- Rash
- Mild bleeding manifestation (e.g., nose or gum bleed, petechiae, or easy bruising)
- Low white cell count
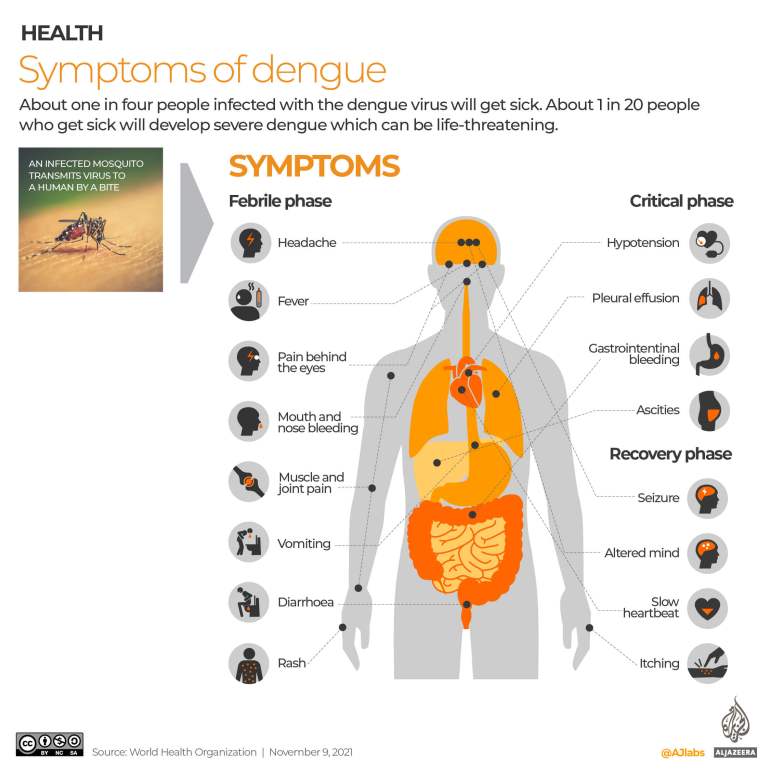
Dengue hemorrhagic fever (DHF), a version of dengue
In addition to concerns about waning immunity, there is also the issue of a more severe form of dengue called dengue hemorrhagic fever (DHF). This form can occur when preexisting antibodies from prior infection make the other strains more dangerous, and more likely that they could develop into a more severe case. See below for symptoms of DHF.
From Science “When Dengue Strikes Twice“:
Most of the more than 50 million people sickened by dengue virus each year develop dengue fever, a weeklong bout of joint and muscle pain. But many who suffer repeat infections have it worse. They come down with dengue hemorrhagic fever and suffer massive internal bleeding and liver damage. Oddly, the virus causing dengue fever comes in four strains, and immunity to one seems to make infection by a second strain more dangerous.
For years, scientists thought that resulting immune system misfire / cell suicide could be a primary cause for DHF, where CD4 cells rapidly destructed following a new infection (versus the more ‘simple’ dengue fever). However, a late 2019 study cleared CD4 T-cells of any wrongdoing. “We found no evidence to support the common dogma that these T cells are responsible for turning a mild infection to a severe one,” said Yuan Tian, PhD, one of the scientists on the study.
The hunt is on still for why DHF is so severe.
Dengue hemorrhagic fever (DHF, or as WHO calls it “severe dengue”) is, as I mentioned above, a more deadly form of dengue first recognized in the 1950s during dengue epidemics in the Philippines and Thailand. In Asia, epidemic DHF expanded from Southeast Asian countries west to India, Sri Lanka, the Maldives, and Pakistan and east to China. (Source)
Today, DHF is in most of Asia, Southeast Asia, and Central and South America, and is a leading cause of hospitalization and death in those regions.
Symptoms of DHF include the above “regular” dengue list plus (source):
- Persistent vomiting
- Severe abdominal pain
- Rapid, shallow breathing
- Blood in vomit or stool
- Bleeding gums or nose
- Vascular leak syndrome
- Skin hemorrhages such as petechiae, purpuric lesions, and ecchymoses.
- Restlessness
- Extreme thirst
People with these severe symptoms should get care right away.
The even more worrisome dengue shock syndrome is defined as dengue hemorrhagic fever plus:
- Weak rapid pulse
- Narrow pulse pressure (less than 20 mm Hg)
- Cold, clammy skin
- Restlessness.
It involves a mostly clinical diagnosis, and is associated with high mortality. (Source)
What to do if you get diagnosed with dengue fever?
Obviously do NOT do what I did and simply not get tested for it. No specific therapy has actually been shown to be effective in the treatment of any of the manifestations of dengue in a randomized controlled trial.
Great, right?
The mainstay of treatment is still careful fluid management (Source). So for treating a dengue infection:
- Get to a hospital and get tested.
- They will likely provide IV hydration and Tylenol.
- Do not use any aspirin or supplements that would thin the blood.
- Do not use NSAIDs either. This means no Advil/Ibuprofen.
- Lots of Nuun tablets and rehydration salts. I use NormaLyte for my electrolytes.
- Following the infection, I found going on a low-inflammation diet or auto-immune protocol diet very helpful. Please see my post about chronic pain for more.
- There is some evidence that mast cell stabilizers and targeting the immune system itself may be helpful, see this study, “Transcriptional Profiling Confirms the Therapeutic Effects of Mast Cell Stabilization in a Dengue Disease Model”
- Avoid any medication that lowers platelet levels. These include Furosemide, NSAIDs (hence the above note about Advil), and some other medications. Best to ask your doctor to confirm.
Yes, you CAN get dengue fever more than once
Yes, as noted above you can get dengue more than once. There are at least four well-characterized serotypes (or strains) of the dengue virus: DENV-1, DENV-2, DENV-3 and DENV-4, and a fifth (DENV-5) was identified in 2015. It is possible to get infected by each serotype, so a person can get dengue up to four times.
Further, over time the existing antibodies for post-infection can wane and people can get the same strain again. It was initially thought that you were immune for life after getting one strain of dengue, but research in recent years has corrected that statement. A study that drew on a large cohort of highly exposed children and adults in Nicaragua (n = 4,478) found evidence of antibody boosting and waning both during and after major dengue and Zika epidemics, suggesting that immunity is not permanent.
As mentioned above, compounding infections may cause something called antibody-dependent enhancement, where the next infection can lead to more severe disease. This means that if someone got infected with dengue, it’s important to try and prevent another infection with a different serotype.
“Long dengue”, genetics, and the lingering effects of an infection
While many friends have come out just fine after a dengue infection, lingering effects are very widely documented.
- Depression and anxiety after the infection wanes (source) – likely due to systemic inflammation. See also this study about anxiety/depression and severity of dengue symptoms.
- Epigenetic changes – much like other severe infections or viruses, geneticists have begun to look at whether your gene composition could affect the severity of the infection or whether it creates sufficient inflammation to “express” genes you may already have and create a genetic cascade.
- Autoimmune marker changes to titers for C3/C4, rheumatoid factor (RF), C-reactive protein (CRP), antinuclear antibodies (ANA), and immune complex (IC). Source.
- According to a survey published in 2020, patients with a history of dengue fever infection are at increased risk of developing leukemia compared with individuals without a history of dengue fever. The findings from this study were reported in Cancer Epidemiology, Biomarkers, & Prevention.
- Developing mast cell dysfunction following dengue fever is also a long-term possibility. Dengue induces increased mast cell activity, for example patients exhibit increased levels of urinary histamine, a major granule product of mast cells. And increased histamine was found to correlate with disease severity. But for some people, their immune systems get “stuck” in that state, post-virally. This is also what scientists think is happening with some Long-Covid patients. I’ve written a long (12,000+ word) page on tackling mast cell issues / increased allergic reactions, here.
- In rare cases, dengue fever can also affect the nervous system and lead to conditions such as myositis, Guillain-Barré syndrome, or hypokalemic paralysis.
- In 2023, the American Centers for Disease Control (CDC) updated their post-viral illness page to list out viruses that can lead to long-term symptoms in the body and included dengue as one of the viruses known to do so.
One significant area of recent research concerns why dengue severity varies so dramatically from person to person. Newer findings point to genetic ancestry as a key factor. Researchers at the University of Pittsburgh and Instituto Aggeu Magalhães in Brazil have for the first time linked the extreme variability in severity of dengue between individuals to the influence of genetic ancestry on inflammatory responses in the skin.
The team exposed human skin explants from donors of European or African ancestry to dengue virus and found that inflammatory responses promoting virus replication and spread are influenced by genetic background, with immune activity increasing with greater European ancestry.
Lead author Dr. Priscila Castanha, an assistant professor of infectious diseases and microbiology at Pitt’s School of Public Health, published her findings in a paper entitled “Genetic ancestry shapes dengue virus infection in human skin explants“, noting that their findings help “explain a longstanding epidemiological puzzle” about why there were reports about the protective effects of African ancestry against severe dengue.
The mechanism, as the team described it, is a kind of “friendly fire”: in samples from donors of European ancestry, myeloid cells mobilized to fight the virus, then themselves became infected, and subsequently migrated out of the skin — similar to how they would travel through the bloodstream into lymph nodes, spreading infection further. In skin samples from donors with higher African ancestry, this cascade was significantly dampened.
The researchers suggest the findings may have implications for how dengue vaccines are designed and assessed, noting that genetic ancestry may affect the way different populations respond to dengue vaccines, which are weakened viruses delivered via the skin. As Dr. Castanha noted, “evolution has made its mark on everyone’s DNA.”
Dengue fever makes mosquitoes bite more
Another interesting factor in dengue’s spread is that new studies have found dengue makes mosquitoes infected with it bite more than mosquitoes who are uninfected. This biting-the-host stuff is called “blood feeding behaviour”, and blood-feeding behavior is a key factor in how mosquitoes spread the disease.
Earlier studies on how dengue virus infection changes the way mosquitoes feed haven’t been that conclusive. But in 2022, researchers took a broader, multidisciplinary approach using a variety of tools like high-resolution video to try and analyze the differences in blood-feeding with mosquitoes who had dengue, and those that did not. A study in PNAS called, Dengue virus infection modifies mosquito blood-feeding behavior to increase transmission to the host, the research team sought to understand transmission to then look at the reasons why these changes happen. And, more promisingly, if they can identify a gene or protein that causes this, they may be able to mitigate against dengue from that angle!
“We found that the dengue virus increases mosquito attraction to the mammalian host and the number of mosquito bites,” says Ashley St. John, associate professor from Duke NUS’ Emerging Infectious Diseases (EID) Programme, and senior coauthor of the study. The higher attraction to the mammalian host increases the chances of the mosquito to bite, while more bites increase the number of transmission events because each bite results in the transmission of the virus.
So essentially: dengue-infected mosquitoes are extra dangerous because they not only carry a pathogen, but spread it more via extra “bites”.
The increase in bites isn’t due to a psychological drive to infect, but rather because the mosquitoes seem to be unable to find blood vessels to feed from as easily, so they bite multiple times to try and get one, thus releasing their dengue-filled saliva into the body more than those mosquitoes who are uninfected (and can find vessels more easily).
The video below shows high-res videos that were then analyzed by computer software, to understand the ways that blood-feeding differed between dengue-mosquitoes and non-dengue mosquitoes.
This is especially an issue a recent research article in Science concluded that mosquitos in Southeast Asia are starting to be resistant to insecticides that countries use to help control the spread of the disease.
Asymptomatic dengue? More common than we thought.
Interestingly, a 2023 study posited that asymptomatic vectors are responsible for a far larger amount of dengue cases than previously realized. The study results found that 8% of areas where people gathered represented more than 50% of all the infections during an outbreak. Of those spaces, 74% infected cases were asymptomatic. These are people who have no symptoms, and continue to live their lives unknowingly infecting mosquitoes that bite them — mosquitos that then go and bite other people and infect them, too.
This study shows that while approaches have historically been reactive (treat people with symptoms; spray / net areas with known outbreaks), we need to also think about containing outbreaks proactively given the role of super spreaders who have no symptoms.
Per Gonzalo Vazquez-Prokopec, one of the study authors:
“Our findings show that any public health intervention that focuses on responding to symptomatic cases of dengue is going to fail to control an outbreak. Symptomatic cases represent only the tip of the iceberg.”
Dengue’s global spread
Per the Center for Disease Control, dengue is endemic throughout the tropics, subtropics, Central and South America, the Caribbean, and Southeast Asia. Presently, it occurs in more than 130 countries worldwide.
Risk now includes the United States, where sporadic local cases have popped up in Florida, Hawaii, and Texas along the border with Mexico, as well as parts of Europe. In 2024, the press in Canada warned people to pay attention to dengue fever when planning a winter vacation,

Although the geographic distribution of dengue is similar to that of malaria, dengue is more of a risk in urban and residential areas than is malaria. As such, the WHO estimates that over 40% of the world’s population live in areas where dengue viruses can be transmitted.
For up to date information of outbreaks in real-time, see the thorough DengueMap. I’ve included a screenshot above.
As of mid-March 2026, the WHO’s Western Pacific Regional Office continued to track active dengue transmission across many countries. In Cambodia, as of epidemiological week 10 of 2026, a total of 4,338 dengue cases including three deaths had been reported — a significant increase from the same period in 2025, when 1,392 cases were reported. Vietnam has seen a two-fold increase in cumulative cases compared to the same period in 2025, with over 27,000 cases reported by early March 2026, and Malaysia had already recorded over 11,000 cases with nine deaths in the first nine weeks of the year.
Singapore’s 2026 serotype profile as of February showed DEN-2 as dominant at 71%, with DEN-3 at 19.4%, DEN-1 at 3.2%, and DEN-4 at 6.5%. This percentage split is worth paying attention to given what we know how serotype shifts can catch populations with low immunity off guard. For the most current surveillance data in the Western Pacific region, the WHO publishes these situation updates regularly, and they are publicly accessible.
In 2026, the Centers for Disease Control and Prevention issued a travel notice of an increase in dengue-infected travellers returning to the USA, especially from those visiting Afghanistan, Bangladesh, Bolivia, Colombia, Cook Islands, Cuba, Guyana, Maldives, Mali, Mauritania, New Caledonia, Pakistan, Samoa, Sudan, Timor-Leste, and Vietnam. Locally-acquired cases are low, but increasing year-over-year. They can be tracked here.

What about Chikungunya and Zika?
Dengue, Zika, and Chikunyunya are all carried by the pesky Aedes aegypti mosquito. As is yellow fever. And of all the mosquitoes out there, this one will happily breed in even the most shallow of water sources. It is unfortunately possible to obtain more than one mosquito-borne disease from the same mosquito.
Aedes aegypti is not the only species to carry the disease, but it is likely the most efficient: It happily takes up residence in human dwellings, biting many people in a row. If it feeds on a sick person, the disease incubates in its belly, then migrates to its salivary glands. It is injected at the next bite.
After six dengue-free decades, Brazil had its first outbreak in 1981. The situation has deteriorated: Last year, 1.6 million people in Brazil were diagnosed with dengue, more than ever before. Chikungunya and Zika have infected hundreds of thousands more.
– Zika Uncontained, Frontline
An American reader living in Manila for many years kindly sent me her story of both dengue and a similar disease called Chikungunya. In her words:
“My experience was horrendous. Very, very similar to what you went through. The reason I am emailing you is to let you know it’s very likely you had both dengue and Chikungunya. The mosquito that carries dengue can also carry Chikungunya. If you get bit by a mosquito carrying both, you can be infected by both dengue and Chikungunya at the same time. This is what happened to me.
While both diseases have similar symptoms, the main difference is dengue can be fatal, Chikungunya isn’t. However, and this is huge — Chikungunya gives you terrible joint and muscle pains. These pains can last up to TWO years! Eventually you will recover completely though. And it can also leave you exhausted. This is all somewhat new – in fact most of us had never heard of Chikungunya before. There was an outbreak in Manila, and there has also been an increasing number of people getting both dengue and Chikungunya at the same time. Most ridiculously, there is not enough information about this.”
So, something to keep in mind if you’re exhibiting symptoms of the kind I mentioned. The rash for Chikungunya is generally on the stomach area, per what I’ve read, and joint pains ongoing. The high fever and joint and muscle pain can last for several months.
The symptoms for both diseases are quite similar, per the CDC, and it has so far been found far outside its usual locations and as far as Texas, Spain (as of August 2015), Mexico, and more, including the Caribbean where it was first detected in the Americas.
The mechanisms of infection of human cells with the virus remain very poorly understood. In September 2019, researchers have identified a protein, four-and-a-half LIM domain protein 1 (FHL1), that is required for the virus to replicate within its target cells. The study shows that FHL1 is a key factor that enables a Chikungunya infection, and allows for a target to potentially develop therapies to treat or preemptively prevent the infection/
Given that the three illnesses offer up similar symptoms, I wanted to devote a part of this post to Zika as well. The virus didn’t just appear in 2016. In fact, it’s been around for a long time. First identified in monkeys in Africa (Uganda to be exact) in the late 1940s, it spread to humans around the early 1950s. The World Health Organization documents the first case at 1952.
Zika’s symptoms are reported as quite mild — far more so than my dengue symptoms noted in this piece.
Per Vox’s Zika Primer,
Zika produces very mild symptoms — rash, headaches, pain in the bones, and fever — that usually show up between three and 12 days after a mosquito bite. These symptoms usually go away within a week, and one in four people don’t even develop any symptoms after being infected with the virus. This means people don’t usually go to the doctor for Zika, and many cases go unnoticed. There’s also no vaccine or treatment for the virus, so doctors just work on controlling and alleviating its symptoms.
Prior to 2013, the disease was not in the Western media, partly because of these mild symptoms and a fairly low mortality rate (compared to, say, malaria or dengue).
However, given its now-establish effects on pregnant women and microcephaly in their fetuses, the disease has become a global concern and opened up debates about women’s rights and contraception in the often-conservative countries that Zika has thus far spread.
Warming planet, moving mosquitoes
As I mentioned in the introduction, with a warming planet, mosquitoes have proliferated, and mosquito-borne illnesses have swept across the globe.
Each year seems to be a banner year for those illnesses, with more and more mainstream news coverage about dengue, chikungunya and Zika.
Said climate scientist Winston Chow from the College of Integrative Studies at Singapore Management University, “constant weather extremes create the perfect breeding conditions for mosquitoes”. With climate changes comes more viral emergencies, and with changing temperatures comes new patterns in global dengue transmissions:

At the time of writing, I have friends in Siem Reap, Cuba, Rio de Janeiro, and Delhi who have dengue at the same time and in very disparate places. One of the hardest symptoms following dengue is the depression and anxiety that follows.
A 2025 study entitled “Disentangling climate’s dual role in dengue dynamics: A multiregion causal analysis study” unveiled new insights into how weather influences the spread of dengue fever as well. The paper identified temperature and rainfall as critical factors driving the global surge in dengue cases and offers actionable strategies for mitigating the disease’s impact, though I will say it’s not a surprise that these two factors play in. We know these mosquitoes love wet climates, and we know climate change is making some places hotter and wetter. (And others drier, of course). The study focused on 16 areas in the Philippines, selected for their diverse climatic conditions, to examine how temperature and rainfall jointly affect dengue dynamics. The most important factor ended up being variation in dry season length: in areas with low variation in dry season length, rainfall tended to flush out stagnant water, reducing mosquito breeding sites and suppressing dengue transmission. But in areas with high variation in dry season length, sporadic rainfall created new breeding sites and weakened the flushing effect, driving an increase in mosquito populations and dengue cases.
The most direct evidence yet of climate change driving a specific dengue outbreak comes from a March 2026 study published in One Earth. Researchers from Stanford University and other institutions traced Peru’s 2023 dengue outbreak directly to cyclone conditions, finding that 60% of cases stemmed from extreme rainfall and heat associated with Cyclone Yaku and a coastal El Niño. Lead author Mallory Harris, now a postdoctoral scholar at the University of Maryland, framed the urgency clearly: the health impacts of climate change “aren’t something we’re waiting for. They’re happening now.”
The numbers behind the paper’s findings are fairly intense: more than 22,000 people likely would not have contracted dengue in the absence of the unusually extreme weather conditions brought by the cyclone. And climate modelling puts this in a longer-term context: extreme precipitation conditions like those seen in 2023 are now 31% more likely in northwestern Peru than they were before industrialization, and when combined with warming temperatures, the probability of climate conditions like those that fueled the 2023 dengue epidemic has nearly tripled.
The study is the first to look at one specific infectious disease outbreak and one specific extreme weather event to rigorously attribute a share of cases to climate, calling it “a big step forward for the methods of the field.”
The practical implication for health systems is that climate-preparedness and epidemic preparedness are now inseparable. The problem is, as we’ve seen in North America, that climate targets are no longer the focus due to increasingly acute geopolitical threats. Still, even smaller actions like targeted mosquito control and vaccination in high-risk urban districts, along with investments in urban flood resilience (like better drainage, sturdier housing, and more reliable water infrastructure) would truly help.
As extreme weather events become more frequent with climate change, scientists stress the need to think strategically and act decisively to prevent mosquito-borne epidemics.
I just don’t know that we will.
New hope: Wolbachia-infected mosquitoes
An interesting solution for the dengue problem: instead of only looking for a vaccine to help prevent the disease, what if the mosquitoes themselves could be defanged? There have been reports of genetically-modified mosquitoes released to test if it lowers the overall dengue cases. Another example of that is a pilot project in Yogakarta, Indonesia, where the mosquitoes are purposely infected with a bacteria commonly found in the insect world.
The bacteria, Wolbachia, was first discovered in 1924 and is found in mosquitoes–just not the Aedes aegypti.
From a June 10, 2021 piece by Ed Yong:
They’ve loaded the mosquitoes with a bacterium called Wolbachia, which prevents them from being infected by dengue viruses. Wolbachia spreads very quickly: If a small number of carrier mosquitoes are released into a neighborhood, almost all of the local insects should be dengue-free within a few months. It’s as if Utarini’s team vaccinated a few individuals against a disease, and soon after the whole population had herd immunity.
and
The team found that just 2.3 percent of feverish people who lived in the Wolbachia release zones had dengue, compared with 9.4 percent in the control areas. Wolbachia also seemed to work against all four dengue serotypes, and reduced the number of dengue hospitalizations by 86 percent.
While the Wolbachia strategy is not immediate, it blocks dengue infections in a few different ways that lead the mosquitoes to be less likely to carry dengue and thus transmit it to people. And we’re already seeing results. In 2023, three cities in Colombia saw a dramatic decline in the incidence of dengue infections after introducing the mosquitoes carrying Wolbachia. In neighbourhoods where the Wolbachia mosquitoes were well established, dengue incidence dropped by 94–97%.
Promising results so far!
Covid-19 and dengue fever
Covid-19’s worldwide spread has further weighed on countries where dengue fever is already an endemic population threat, primarily in tropical and subtropical regions of the world. But as discussed here, dengue is spreading more widely, now, and people do need to be aware of how these two viruses interact in the body.
Both Covid and dengue share some symptoms, and this has led to diagnostic challenges. Moreover, cross-reactivity of the body’s immune response to each of these infections is something on scientists’ radar, since the concern is that pre-existing dengue antibodies might affect the body’s ability to clear Covid, or create something called antibody-dependent enhancement (ADE), which is where existing antibodies facilitate a virus’ entry into cells and encourage replication.

First raised in 2022, it was hypothesized that dengue antibodies could either help cushion a Covid infection (making it milder), or may create ADE and thus potentially worsen outcomes.
A January 2026 study found the opposite: that antibodies to Covid acquired from natural infection in humans or through experimental immunization in animals were cross-reactive with DENV-2 strain of dengue, and had the potential to enhance DENV-2 infection in cells. In plain terms, Covid infection can lead to elevated risks and symptoms in subsequent dengue infections.
A November 2025 paper published in The Lancet, “Risk of dengue following prior SARS-CoV-2 infection: a population-based cohort study“, also found that for people who had Covid, it created an increased risk of DENV-infection (primarily DENV3) for up to 300 days. It also notes that while this is “not irrefutable proof for possible antibody-dependent enhancement of DENV3-infection post-Omicron COVID-19, given modest effect sizes, future studies can shed more light on potential interactions.”
But what about the inverse?
An earlier 2025 paper entitled “Effects of Recent Prior Dengue Infection on Risk and Severity of Subsequent SARS-CoV-2 Infection: A Retrospective Cohort Study” came to similar conclusions, noting:
Increased risk of SARS-CoV-2 infection and adverse COVID-19 outcomes were observed following preceding dengue infection in a national population-based cohort of adult Singaporeans. This observation is of significance in tropical countries with overlapping dengue and COVID-19 outbreaks.
Regardless of what further results show, it’s important to make sure you’re testing appropriately to know whether it’s dengue, Covid, or something else so that you can treat appropriately. If these study directions bear out, I fear that as reinfections of Covid stack up, it will only worsen dengue outcomes.

Dengue and COVID-19: two sides of the same coin
Dengue led to Mast Cell Activation Syndrome (MCAS) for me
While I addressed the concept of “long dengue” above, for me the fatigue, the hair loss, the joint pain, and the depression and anxiety I spoke of in my piece about chronic pain was different to my friends’ experiences with dengue.
Many friends had an infection and got over it just fine. However, with an existing autoimmune condition (celiac), the infection was really difficult. For others, they had a few months of problems but are fine now.
I never got “back to normal”.
It has been over a decade, and the infection plus my stubborn unwillingness to stop traveling and rest longer-term led to an inflammatory immune condition called mast cell activation syndrome, which is similar in symptoms to long covid descriptions. I even had what people are calling “covid toe” for years.
At the time of my infection in 2013, I thought I’d just take time to get better. Instead, my immune system got more and more reactive and in 2017, I had a diagnostic lumbar puncture done that led to a spinal CSF Leak that is still ongoing.
Like Covid for some people, dengue changed my body and immune system and I became reactive to environmental and other triggers that I’d spent 40 years of my life having no issues with. I now have many new reactions to foods, smells, even the sun. I go into anaphylaxis on the regular. Vascular changes abound, and I have become intolerant to hot or very cold weather, my toes go blue in the shower, my circulation is affected by environmental triggers as well, to the point where I lose feeling in my extremities as they swell when exposed to them. This is especially bad during pollen season, and ‘smoke season’ (either due to wildfires, or heating homes with woodsmoke which is common in Quebec, Canada.
My story is part of why I wanted to write this page: so everyone could understand that especially with existing Covid antibodies, you want to try and avoid dengue fever.
Additional reading about dengue fever:
- Interesting article from the Smithsonian Magazine, about how the isolation of a single protein could help develop a vaccine for dengue. This is a different vaccine than the Sanofi one that is in development and testing stages, since it targets a protein that, if this piece is correct, could help protect against all of the dengue strains.
- A 2019 piece in the New York Times, “The Mosquitoes are Coming for Us” goes into history as well. “The mosquito and her diseases have accompanied traders, travelers, soldiers and settlers (and their captive African slaves) around the world and have been far more lethal than any manufactured weapons or inventions.”
- A 2019 article about how mosquitoes “slaughtered our ancestors and derailed history” in the New Yorker. “Along with smallpox and influenza, mosquito-borne diseases led, by Winegard’s estimate, to the deaths of ninety-five million indigenous inhabitants of the Americas, from a pre-contact population of about a hundred million.“
- For many years, there was no safe vaccine available. Previously, the Sanofi vaccine, Dengvaxia, rolled out to very problematic results. In December 2022, though, a dengue vaccine developed by Japanese drugmaker Takeda Pharmaceutical Co was authorised for use in the European Union. The vaccine, branded QDENGA, is authorized for use in those aged 4 and older to prevent any of the four serotypes of dengue. Takeda’s vaccine is based on a Dengue 2 virus, with DNA from the other three serotypes added in so it protects against all serotypes. Data from Takeda’s main trial showed that the vaccine induces immune responses (varied ones, depending on the person’s immune system) against all four dengue types. The issue, now that the 2023 immunity study from Nicaragua has been published, is how long these antibodies will last.
- In 2023, pharmaceutical company Janssen shared promising data on JNJ-1802, an antiviral drug that is aimed at preventing dengue and is taken orally (in pill form). While likely not feasible to give to everyone in dengue-filled areas, should these data pan out in further trials, it will be an option for travelers much like anti-malarials.
